|
9/20/2023 0 Comments Second order half lifeAnd as a result of this, it's going to be constant throughout the whole reaction. So that means half life does not depend on the initial concentration of our reactant. We don't see initial concentration of our reactant anywhere. Now, what can we say about this half life equation and concentration? Well, if we take a look, half life equals Ln two over K. And verse T of course is time when it's T. And here we'd say since it's dealing with first order it would be units of time. In this case we're going to say K is our rate constant. Here we're gonna say Ln two is our constant. 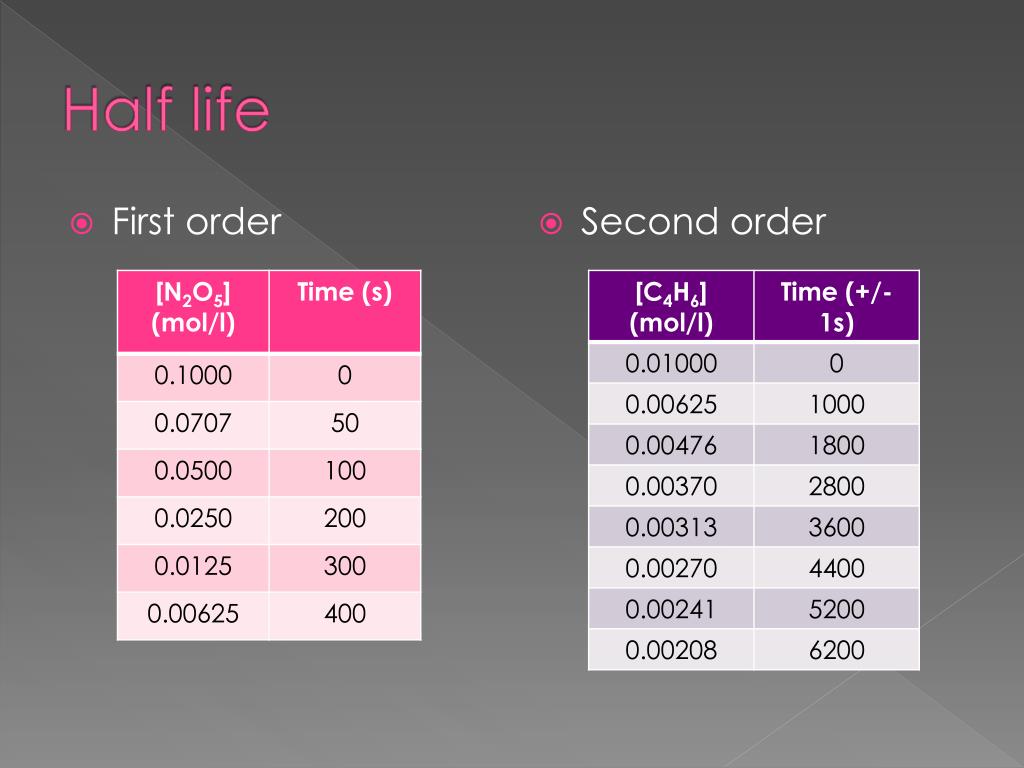
You may see on your formula sheet instead of Ln two, you may see. Now, if you plug in Ln two Ln two in your calculator, you're gonna get 20.693 And it will still be over K. So for reactions with first order, we use the following equation and that is half black equals Ln two over K. Now when it comes to first order in half life first realized that all radioactive processes follow a first order rate law. So this comes out to be 8.52 times to the three seconds. The polarities will cancel out and our time our half life will be in seconds. We have the initial concentration of our reactant in terms of ammonia divided by two times our rate constant here, malaria, t times seconds inverse. So this becomes a simple plug and chug type of question. So here that means that half life equals the initial concentration of your reactant divided by two K. So the fact that its polarity times seconds in verse tells us this is a zero order reaction. Because remember for a zero order reaction, the units for your rate constants are polarity times time inverse. Alright, so here we know that this is a zero order reaction. 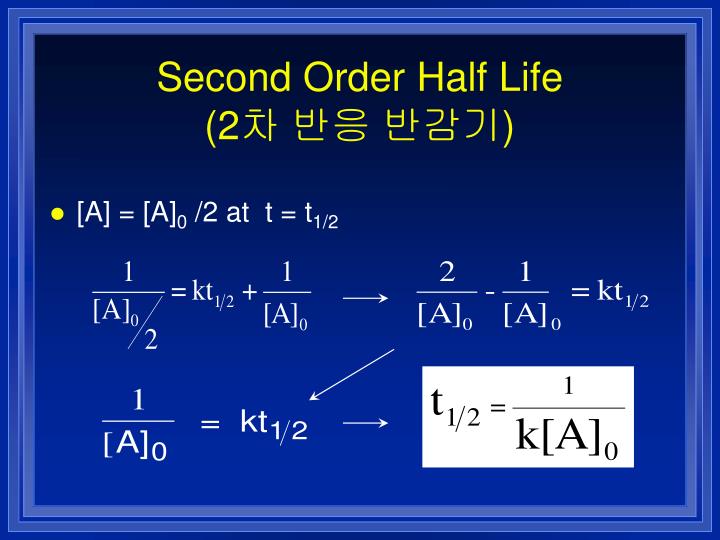
Now calculate the half life for the reaction, where the initial concentration of ammonia equals 2.47 times 10 to the negative two moles per liter. The reverse haber reaction has a rate constant of 1.45 times 10 to negative six polarity times seconds inverse at 25 degrees Celsius. Alright, so keep in mind that this is the half life equation for a zero order process, where half life equals initial concentration of your reactant divided by two K. So here that's why half life is on the line and time is on the X axis, respectively. And remember when we're doing a plot it's always of Y versus X. 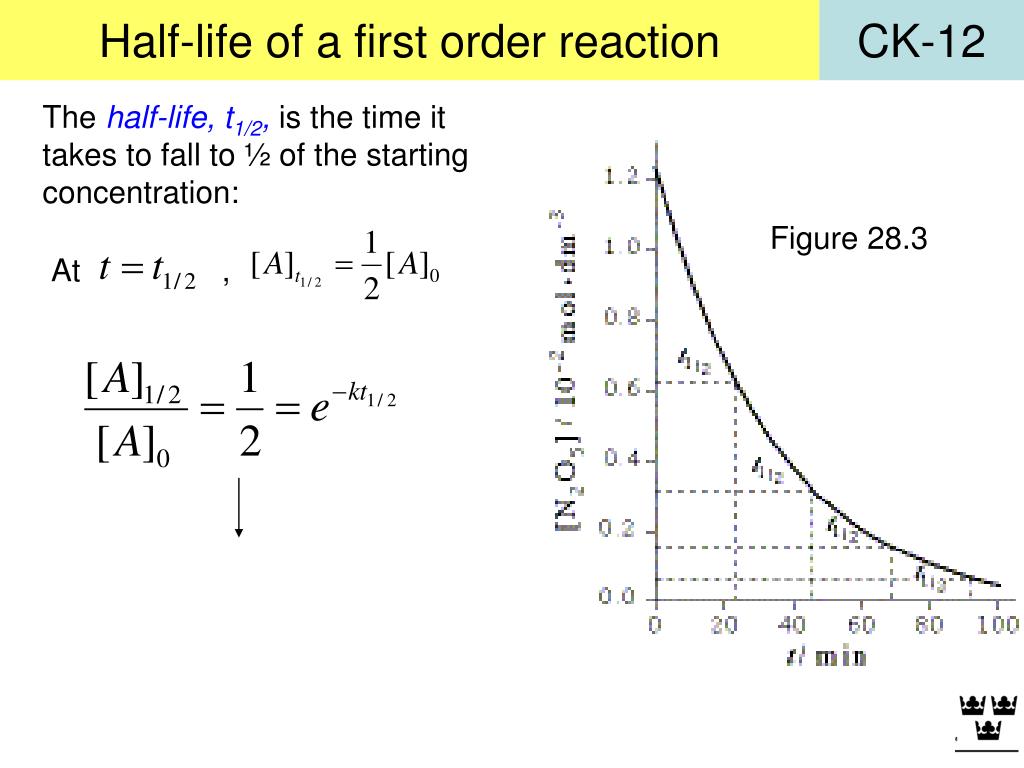
And here remember we have our half life as the Y axis and our time as the X. Okay, so we see it with a negative slope decreasing over time. So what would that look like graphically we'd say that as time progresses that our time gets shorter and shorter. That means that half life starts to drop the smaller half life, the less time it takes for you to lose half of your initial amount now here um the time gets shorter over time. Because I think about it if this number starts off at let's say 100 100 divided by two K. Well here we're going to say that since half life is part of the equation, we're going to say half life does depend on the initial concentration of our reactant and it gets shorter as the concentration decreases. Now if we take a look at this we'd say what happens to half life? If our initial concentration gets blank as concentration decreases. But when we have t half that really stands for half life. Remember for for um for zero order processes we'd say that its units of polarity times time, inverse here, T in this case is time. Here a sub zero is again our initial reacting concentration K equals our rate constant. Now if we're talking about zero order, we're gonna say for zero order reactions, we use the following equation for half life here half life equals the initial concentration of our reactant divided by two times K. Remember we have zeroth order, first order and second order. Now here the way half by function really depends on the order. Now half life is just the time it takes to lose half of a reacting to decay or decomposed in a certain time period.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
